News
Keep updated with the latest MDPP news via our newsletter.
Fantastic news for Medical Device Partnering Program, Medical Device Research Institute and South Australia’s health and medical research sector! 🎉
We’re proud to share that our Director, Prof Karen Reynolds, has been appointed to the National Health and Medical Research Council (NHMRC) - MRFF Industry, Philanthropy and Commercialisation Committee (2024-2027)!
Karen joins an outstanding group of leaders, including A/Prof Jiawen Li and Kylie Sproston FTSE GAICD, in shaping the future of Australian medical research and industry collaboration.
This is a well-deserved recognition of Karen’s leadership in bridging research, industry, and healthcare to drive real-world impact.
Congratulations, Karen! 👏
Read more here: bit.ly/4gNmWNs

An Australian clinical surgeon has collaborated with the Medical Device Partnering Program (MDPP) at Flinders University to develop a novel catheter for safely navigating the intricate curves and contours of the male urinary tract.
Urological surgeon Associate Professor Ian Middleton developed "DeflectaCath," a silicone catheter equipped with a unique deflection mechanism in its tip, to simplify the insertion procedure for clinicians and improve the health outcomes for patients.
DeflectaCath provides a safe and comfortable catheterisation solution for men managing health conditions or undergoing medical procedures. It allows practitioners to manoeuvre the catheter through the length and curvature of the urethra, effectively minimising the risk of potential complications.
A/Prof Middleton said it was exciting working with the MDPP team to prototype a standout medical device that improves the catheterisation experience for both healthcare professionals and their patients.
“Working closely with the MDPP team has allowed us to concentrate on innovating the design of DeflectaCath, ensuring an easy and intuitive insertion and placement for medical staff while reducing the associated health risks of urethral trauma for patients,” said A/Prof Middleton.
The unique deflection mechanism in DeflectaCath's tip enhances device maneuverability, minimising the chances of urethral damage or the formation of false passages that can occur from urethral wall perforation. This advancement is especially critical with studies reporting that catheterisation poses challenges in around one in five cases.
“We’re extremely excited to have developed an efficient and effective catheterisation alternative which is unlike any of the current catheters being used by clinicians,” said A/Prof Middleton. “Thanks to the MDPP team, DeflectaCath now includes manual control, allowing medical practitioners to navigate a patient's anatomy throughout the entire procedure. This capability represents a game-changing advancement for both practitioners and patients,”
Professor Karen Reynolds, director of MDPP, highlighted that DeflectaCath's potential transformative impact on healthcare delivery marks a new era of enhanced patient outcomes and improved quality of care in urology.
“Our team takes pride in supporting innovators like Ian to find novel solutions and DeflectaCath represents a significant breakthrough in urological procedures,” said Prof Reynolds. “Helping to revolutionise the catheterisation process reflects our steadfast commitment to innovation and advancing patient care and outcomes.”
This project involved resources from the Medical Device Research Institute (MDRI) at Flinders University, the Australian National Fabrication Facility (ANFF) and the team at MDPP provided 250 hours towards proof-of-concept research, prototyping and product validation. Fostering collaborations between researchers, industry, end-users, and government to develop medical technologies with global market potential, the MDPP is an ideas incubator. Do you have an innovative medtech idea? Submit your idea through our website and a member of the team will get in touch. For any other questions please contact us on 08 8201 5029 or email This email address is being protected from spambots. You need JavaScript enabled to view it..

Photo: A transparent model of the urethral anatomy has been produced to demonstrate how the deflection mechanism can allow the catheter to bypass a false passage and prevent new trauma to the urethra.
We are happy to announce that the SA State Government will continue to support the Medical Device Partnering Program so we can keep supporting early-stage medtech innovators in Australia. The South Australian Government is investing a further $900,000 to support the continuation of the Medical Device Partnering Program (MDPP).
An initiative of Flinders University's Medical Device Research Institute, the MDPP works to foster collaboration between researchers, industry and end-users to create medical technologies where there is both identified clinical need and viable market opportunity.
Read the news release in full here: https://lnkd.in/gNXUYEvR
Established in 2008, the MDPP supports companies with up to 250 hours of technical expertise, fast-tracking their research and development process and allowing participants to explore opportunities and resolve technical problems.

We're hiring!
We’re looking for a new Innovations Manager at Medical Device Partnering Program!
The Innovations Manager will work with applicants to the Medical Device Partnering Program, assessing the viability of new ideas and connecting them with the right expertise. Perfect for anyone who has an interest in medtech commercialisation, a scientific background and enjoys learning about things they never expected to need to know! You get to work with a great team and an amazing array of innovators from around the country, help create new start up companies and help them in their journey!
Applications close: 19 May 2024.
-
Fixed term until June 2025 | Full-Time (Part-Time Considered)
-
Higher Education Officer Level 8| $106,842 - $118,281 p.a.
-
Location: Tonsley / Kaurna Country
For more information and to apply visit -
https://flinders.wd3.myworkdayjobs.com/en-US/flindersemployment/details/Innovations-ManagerJR0000010626
The catheterisation of male patients is difficult and frustrating for urologists and, after years of contemplation, busy surgeon Ian Middleton decided he would help tackle the situation. Partnering with the Medical Device Partnering Program enabled Ian to develop a working prototype of his medical device and he is now seeking a catheter manufacturer for a pathway to production.
In his latest publication, Ian discusses the arduous process of patent acquisition - which is vital for innovation – and expresses his appreciation to the MDPP team for supporting and guiding him through the commercialisation pipeline.
Thanks for your kind words, Ian. We have enjoyed working with you!

Click here to read the paper: MiddletonPaper.PDF
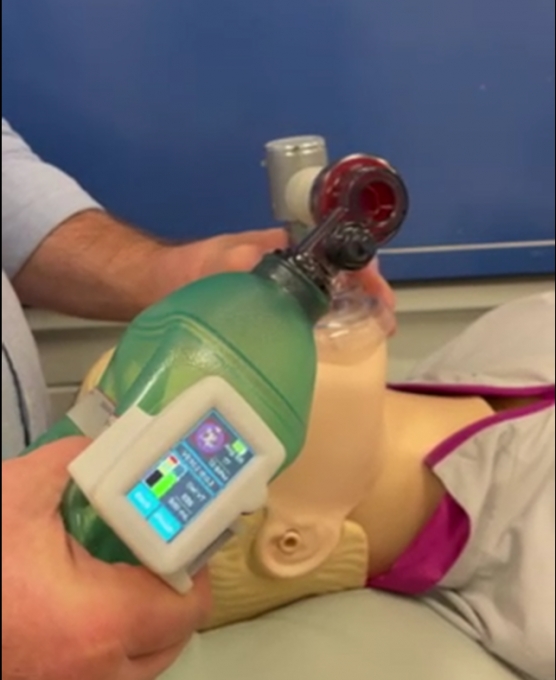
Ventilation management during life-saving resuscitation is being transformed by a local start-up, Abtulus®, who have partnered with the Medical Device Partnering Program (MDPP) at Flinders University to develop a novel Bag Valve Mask (BVM) guidance system.
Engineered by Dr Siavash Noor (Ahmadi Noorbakhsh) of Abtulus®, VentiWatch is a practical, simple and reusable ventilation management solution supporting medical professionals and first responders with the delivery of precise and efficient ventilation during resuscitation.
The cutting-edge sensor technology eliminates the common challenges associated with using BVMs, including inaccurate placement and incorrect technique of ventilation, which can cause further complications like delivering less than needed ventilations (hypoventilation) or pushing too much air into the lungs (hyperventilation) of patients in critical conditions.
The joint effort to advance CPR ventilation management was achieved with the MDPP team crafting a custom coil and sensing circuit, complete with sophisticated processing algorithm, to provide accurate estimates of air volume delivery and ventilation timing with real-time feedback.
Placed on the outside of the BVM, VentiWatch connects to the ventilation management device which features a user-friendly interface, colour LCD screen, navigation switch, audible alarms, a micro-USB port and a rechargeable lithium-ion battery.
First responders can then inflate a patient’s lungs with a squeeze of the BVM to obtain the perfect volume and timing for a successful resuscitation and VentiWatch’s continuous ventilation monitoring system then enables the first responders to safely transfer high-risk patients to hospital emergency departments.
 “The expertise, resources, and support provided by the MDPP team throughout the program have propelled my device and company to a whole new level,” remarked Dr Noor, Founder and CEO of Abtulus and inventor of VentiWatch.
“The expertise, resources, and support provided by the MDPP team throughout the program have propelled my device and company to a whole new level,” remarked Dr Noor, Founder and CEO of Abtulus and inventor of VentiWatch.
“Overall, working with MDPP has been an enriching experience. I look forward to a continued partnership as we bring this life-saving technology to the world.”
Professor Karen Reynolds, Director of MDPP, says VentiWatch has evolved the BVM system into a sophisticated tool with the potential to revolutionise CPR ventilation management.
“Through every stage of the project, our team of experts have consulted with Dr Noor, refining and optimising his initial design, while preserving its simplicity and reusability,” said Prof Reynolds.
“The BVM guidance system we have built for Dr Noor is the optimal choice in ventilation management support for first responders attending life-threatening emergencies.
We are extremely proud to be supporting the development of a novel medical device designed to deliver a solution that ensures optimal patient care during critical resuscitation scenarios.
The MDPP supports the development of new, high-tech medical devices through facilitating collaboration between researchers, industry, end- users and government, and undertaking rapid research projects that demonstrate proof of concept and de-risk ideas.

Sorry you missed this great event. Keep an eye on our website or Twitter for upcoming events.
You are invited to a seminar by guest presenter Prof Peter Hunter from University of Auckland
When: 10am on Monday 4th March
Where: Flinders University, 1284 South Road, Tonsley
Presentation title: A physics-based physiology approach for developing the virtual human twin
Professor Peter Hunter is a Distinguished Professor and founder of the Auckland Bioengineering Institute at the University of Auckland, co-Director of Computational Physiology at Oxford University and holds honorary or visiting Professorships at a number of Universities around the world. He is on the scientific advisory boards of a number of Research Institutes in Europe, the US and the Asia-Pacific region.
Professor Hunter's major research interests are on modelling many aspects of the human body using specially developed computational algorithms and an anatomically and biophysically based approach which incorporates detailed anatomical and microstructural measurements and material properties into the continuum models.
Please register via Eventbrite if you would like to attend.
The event will be livestreamed for those unable to attend in person.